 
The microscopic kinetic energy is sometimes called thermal energy, which is proportional to temperature. Internal energy of a substance is the sum of the potential energy arising from chemical bonds of atoms and electrons and the sum of the kinetic energy of the atoms and molecules. M indicates mass, and Q indicates heat.īefore proceeding, we must define internal energy. 1 – Three types of thermodynamic systems. Heat transfer is positive when heat is exchanged from the surroundings to the system.įig. Energy in the form of heat might enter or leave the system across the system boundaries.We neglect potential energy and kinetic energy changes within the system.The mass that enters or exits the system also does work-sometimes called flow work or pressure work. Work is positive when the surroundings do work on the system (i.e., the system contracts). Thus, work can be done by the system on the surroundings or vice versa. The boundaries of the system can expand or contract. The only types of work that are present are expansion/compression of the system and flow work.This is often a good assumption when the fluid is not moving near the speed of sound, the change in height over the system is not large, or the system temperature variations are not large. We neglect kinetic and potential energy carried by the mass. Mass can carry internal energy into or out of the system.The mass flow rate into the system is positive, whereas flow rates out of the system to the surroundings are negative. Mass flows into or out of the system along one boundary of the system.We make the following assumptions and definitions: 1, an open system allows mass and energy to flow into or out of the system. No third law was injured or killed in this comment, names were withheld to protect the guilty.We begin with the first law of thermodynamics applied to an open thermodynamic system. Suction is the pushing towards you, and pushing is the pushing away from you. 
In other words, at the initial starting point in the middle of nowhere in the universe, at the final time of reaction, if you were to go where the initial reaction occurred, you would find zero mass and or energy and not your rocket with the gas and oxygen sucked out instantly by the abstract mislead concept of a vacuum as nothing sucks. The math you requested: (Rocket mass and Energy) - (gas mass and Energy) = 0. The flat Earth Society claim to have members all over the "globe". Lastly, if you don't want to hear Newton's third law then there is no sense of talking about a spherical world. There is nothing in our universe that reacts instantly as that would get a speeding ticket for going faster than the speed of light speed limit,not to include the things that are already travelling faster than the speed of light as they would get a slowing ticket for going past, or below the speed of light, starting at the Planck scale. Nice question, but incomplete, you forgot to add gravity, which there is no place in our universe with zero gravity as gravity is a field, also, the addition of time. If anyone can answer how propulsion happens in the ever expanding vacuum of space using rocket technology with math that does not conflict with the PV work formula I would like to hear the answer. free expansion therefore no gas will acculmate at the nozzle to push off or react upon as I have heard some space agencies claim. 
If burnt gasses come out of the nozzle (action) they will be obsorbed by the infinite vacuum of space A.K.A. 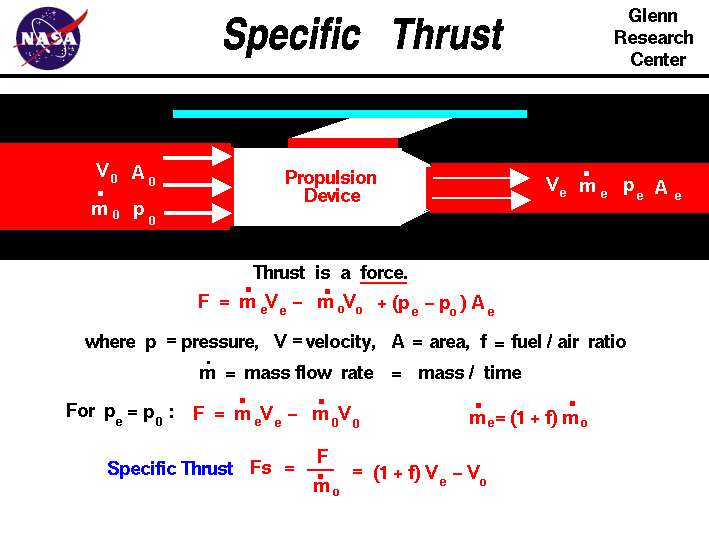
If fuel and oxygen are mixed in the combustion chamber and somehow ignited the laws of free expansion eliminate any work from being performed and therefore no thrust happens because there is no external resistance/pressure to get an opposite and equal reaction from. If I am wrong than so is the P V work formula, and please don't throw Newton's third law as an answer, without explaining where the reaction comes from because pushing off nothing makes no sense, and in a vacuum the gasses are subject to free expansion. My conclusion is rockets can not produce thrust in the supposed vacuum of space, because there is no resistance/pressure to work on. If oxygen and a combustible fuel are mixed in a combustion chamber that is in equilibrium with the ever expanding vacuum of space how can any work be done? W = P ext is 0 X any number for change in volume = zero, because any number muitiplied by zero is zero, therefore 0 work is performed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
